10-Q: Quarterly report pursuant to Section 13 or 15(d)
Published on February 12, 2024
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
______________________________________
FORM
______________________________________
(Mark One)
QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the quarterly period ended
or
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number:
______________________________________
(Exact name of registrant as specified in its charter)
______________________________________
Québec, |
|
(State or other jurisdiction of |
(I.R.S. Employer |
(Address of principal executive offices, including zip code)
(Registrant’s telephone number, including area code)
(Former name, former address, and former fiscal year, if changed since last report)
______________________________________
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer |
☐ |
|
Accelerated filer |
☐ |
|
☒ |
|
Smaller reporting company |
|
||
Emerging growth company |
|
|
|
|
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes
The number of outstanding common shares of the registrant, no par value per share, as of February 9, 2024, was
ACASTI PHARMA INC.
QUARTERLY REPORT ON FORM 10-Q
For the Quarter Ended December 31, 2023
Table of Contents
|
|
Page |
|
||
4 |
||
|
|
|
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
19 |
|
|
|
|
Item 3. |
37 |
|
|
|
|
Item 4. |
37 |
|
|
|
|
|
||
37 |
||
|
|
|
38 |
||
|
|
|
38 |
||
|
|
|
38 |
||
|
|
|
38 |
||
|
|
|
38 |
||
|
|
|
38 |
||
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This quarterly report contains information that may be forward-looking statements within the meaning of Canadian securities laws or forward-looking statements within the meaning of U.S. federal securities laws, and we refer to such statements in this quarterly report as forward-looking statements. Forward- looking statements can be identified by the use of terms such as “may”, “will”, “should”, “expect”, “plan”, “anticipate”, “believe”, “intend”, “estimate”, “predict”, “potential”, “continue” or other similar expressions concerning matters that are not statements about the present or historical facts.
Although the forward-looking statements in this quarterly report are based upon what we believe are reasonable assumptions, you should not place undue reliance on those forward-looking statements since actual results may vary materially from them.
In addition, the forward-looking statements in this quarterly report are subject to a number of known and unknown risks, uncertainties and other factors, many of which are beyond our control, that could cause our actual results and developments to differ materially from those that are disclosed in or implied by the forward-looking statements, including, among others:
2
All of the forward-looking statements in this quarterly report are qualified by this cautionary statement. There can be no guarantee that the results or developments that we anticipate will be realized or, even if substantially realized, that they will have the consequences or effects on our business, financial condition, or results of operations that we anticipate. As a result, you should not place undue reliance on these forward-looking statements. Except as required by applicable law, we do not undertake to update or amend any forward-looking statements, whether as a result of new information, future events or otherwise. All forward-looking statements are made as of the date of this quarterly report.
We express all amounts in this quarterly report in U.S. dollars, except where otherwise indicated. References to “$” are to U.S. dollars and references to “CAD$” are to Canadian dollars.
Except as otherwise indicated, references in this quarterly report to “Acasti,” “the Corporation,” “we,” “us” and “our” refer to Acasti Pharma Inc. and its consolidated subsidiaries.
3
PART I. FINANCIAL INFORMATION
Item 1: Financial Information
Unaudited Condensed Consolidated Interim Financial Statements
4
ACASTI PHARMA INC.
Condensed Consolidated Interim Balance Sheets
(Unaudited)
|
|
December 31, |
|
March 31, |
(Expressed in thousands except share data) |
|
$ |
|
$ |
Assets |
|
|
|
|
|
|
|
|
|
Current assets: |
|
|
|
|
Cash and cash equivalents |
|
|
||
Short-term investments |
|
|
||
Receivables |
|
|
||
Prepaid expenses |
|
|
||
Total current assets |
|
|
||
|
|
|
|
|
Operating lease right of use asset |
|
|
||
Equipment |
|
|
||
Intangible assets |
|
|
||
Goodwill |
|
|
||
Total assets |
|
|
||
|
|
|
|
|
Liabilities and Shareholders’ equity |
|
|
|
|
Current liabilities: |
|
|
|
|
Trade and other payables |
|
|
||
Operating lease liability |
|
|
||
Total current liabilities |
|
|
||
|
|
|
|
|
Derivative warrant liabilities |
|
|
— |
|
Operating lease liability |
|
|
||
Deferred tax liability |
|
|
||
Total liabilities |
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Shareholders’ equity: |
|
|
|
|
Class A common shares, |
|
|
||
Class B, C, D and E common shares, |
|
|
||
Additional paid-in capital |
|
|
||
Accumulated other comprehensive loss |
|
( |
|
( |
Accumulated deficit |
|
( |
|
( |
Total shareholders' equity |
|
|
||
|
|
|
|
|
Total liabilities and shareholders’ equity |
|
|
See accompanying notes to unaudited interim consolidated financial statements.
5
ACASTI PHARMA INC.
Condensed Consolidated Interim Statements of Loss and Comprehensive Loss
(Unaudited)
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||
|
|
December 31, |
|
|
December 31, |
|
|
December 31, |
|
|
December 31, |
|
||||
(Expressed in thousands, except share and per share data) |
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Operating expenses |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Research and development expenses, net of government assistance |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
General and administrative expenses |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Sales and marketing |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
Restructuring cost |
|
|
|
|
|
|
|
|
( |
) |
|
|
|
|||
Loss from operating activities |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Foreign exchange gain (loss) |
|
|
|
|
|
|
|
|
( |
) |
|
|
( |
) |
||
Change in fair value of derivative warrant liabilities |
|
|
|
|
|
|
|
|
( |
) |
|
|
|
|||
Interest income and other expense, net |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Total other income (expense), net |
|
|
|
|
|
|
|
|
( |
) |
|
|
|
|||
Loss before income tax recovery |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Income tax recovery |
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Net loss and total comprehensive loss |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Basic and diluted loss per share |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
Weighted-average number of shares outstanding |
|
|
|
|
|
|
|
|
|
|
|
|
||||
See accompanying notes to unaudited interim consolidated financial statements
6
ACASTI PARMA INC.
Condensed Consolidated Interim Statements of Shareholders' Equity
(Unaudited)
|
|
Common Shares |
|
|
|
|
|
|
|
|
||
(Expressed in thousands except share data) |
|
Number |
|
Dollar |
|
Additional |
|
Accumulated |
|
Accumulated deficit |
|
Total stockholders' equity |
|
|
|
|
$ |
|
$ |
|
$ |
|
$ |
|
$ |
Balance, March 31, 2023 |
|
|
|
|
( |
|
( |
|
||||
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
— |
|
||
Balance at June 30, 2023 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common shares and pre-funded warrants through private placement, net of offering costs |
|
|
|
|
— |
|
— |
|
||||
Issuance of common shares upon the exercise of stock options |
|
|
— |
|
|
— |
|
— |
|
|||
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
— |
|
||
Balance at September 30, 2023 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
— |
|
||
Balance at December 31, 2023 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Shares |
|
|
|
|
|
|
|
|
||
(Expressed in thousands except for share data) |
|
Number |
|
Dollar |
|
Additional |
|
Accumulated |
|
Accumulated deficit |
|
Total stockholders' equity |
|
|
|
|
$ |
|
$ |
|
$ |
|
$ |
|
$ |
Balance, March 31, 2022 |
|
|
|
|
( |
|
( |
|
||||
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Cumulative translation adjustment |
|
— |
|
— |
|
— |
|
( |
|
— |
|
( |
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
— |
|
||
Net proceeds from shares issued under the at-the-market (ATM) program |
|
|
|
— |
|
— |
|
— |
|
|||
Balance at June 30, 2022 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Cumulative translation adjustment |
|
— |
|
— |
|
— |
|
( |
|
— |
|
( |
Net proceeds from shares issued under the at-the-market (ATM) program |
|
|
|
— |
|
— |
|
— |
|
|||
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
— |
|
||
Balance at September 30, 2022 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss and total comprehensive loss for the period |
|
— |
|
— |
|
— |
|
— |
|
( |
|
( |
Cumulative translation adjustment |
|
— |
|
— |
|
— |
|
|
— |
|
||
Stock-based compensation |
|
— |
|
— |
|
|
— |
|
|
|
||
Balance at December 31, 2022 |
|
|
|
|
( |
|
( |
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
7
ACASTI PHARMA INC.
Condensed Consolidated Interim Statements of Cash Flows
(Unaudited)
|
|
Nine months ended |
|
|||||
|
|
December 31, |
|
|
December 31, |
|
||
(Expressed in thousands) |
|
$ |
|
|
$ |
|
||
Cash flows used in operating activities: |
|
|
|
|
|
|
||
Net loss for the period |
|
|
( |
) |
|
|
( |
) |
Adjustments: |
|
|
|
|
|
|
||
Depreciation of equipment |
|
|
|
|
|
|
||
Gain on sale of equipment |
|
|
( |
) |
|
|
|
|
Stock-based compensation |
|
|
|
|
|
|
||
Change in fair value of warrant liabilities |
|
|
|
|
|
( |
) |
|
Income tax recovery |
|
|
( |
) |
|
|
( |
) |
Unrealized foreign exchange (gain) loss |
|
|
|
|
|
( |
) |
|
Write-off of equipment |
|
|
|
|
|
|
||
Changes in operating assets and liabilities: |
|
|
|
|
|
|
||
Receivables |
|
|
( |
) |
|
|
( |
) |
Prepaid expenses |
|
|
( |
) |
|
|
( |
) |
Trade and other payables |
|
|
( |
) |
|
|
|
|
Operating lease right of use asset |
|
|
( |
) |
|
|
|
|
Net cash used in operating activities |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
||
Cash flows from investing activities: |
|
|
|
|
|
|
||
Acquisition of equipment |
|
|
|
|
|
( |
) |
|
Proceeds from sale of equipment |
|
|
|
|
|
|
||
Acquisition of short-term investments |
|
|
( |
) |
|
|
( |
) |
Maturity of short-term investments |
|
|
|
|
|
|
||
Net cash (used in) provided by investing activities |
|
|
( |
) |
|
|
|
|
|
|
|
|
|
|
|
||
Cash flows from financing activities: |
|
|
|
|
|
|
||
Net proceeds from issuance of common shares and warrants from private placement |
|
|
|
|
|
|
||
Proceeds from issuance of common shares from exercise of stock options |
|
|
|
|
|
|
||
Net proceeds from shares issuance under the at-the-market (ATM) program |
|
|
|
|
|
|
||
Net cash provided by financing activities |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
Effect of exchange rate fluctuations on cash and cash equivalents |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
Net decrease in cash and cash equivalents |
|
|
( |
) |
|
|
( |
) |
|
|
|
|
|
|
|
||
Cash and cash equivalents, beginning of period |
|
|
|
|
|
|
||
Cash and cash equivalents, end of period |
|
|
|
|
|
|
||
|
|
|
|
|
|
|
||
Cash and cash equivalents are comprised of: |
|
|
|
|
|
|
||
Cash |
|
|
|
|
|
|
||
Cash equivalents |
|
|
|
|
|
|
||
See accompanying notes to unaudited interim consolidated financial statements.
8
ACASTI PHARMA INC.
Notes to Condensed Interim Consolidated Financial Statements
(Unaudited)
(Expressed in thousands except share and per share data)
1. Nature of operation
Acasti Pharma Inc. (“Acasti” or the “Corporation”) is incorporated under the Business Corporations Act (Québec) (formerly Part 1A of the Companies Act (Québec)). The Corporation is domiciled in Canada and its registered office is located at 2572 boul. Daniel-Johnson, 2nd Floor Laval, Québec, Canada H7T 2R3.
The Corporation’s Class A common shares, no par value per share (“Common Shares”), are listed on the Nasdaq Capital Market (the “Nasdaq”) and, through March 27, 2023, the Corporation's Common Shares were also listed on the TSX Venture Exchange (“TSXV”), in each case, under the symbol “ACST”. On March 13, 2023, the Corporation received approval to voluntarily delist from the TSXV. Effective as at the close of trading on March 27, 2023, the Corporation's Common Shares are no longer listed and posted for trading on the TSXV.
In August 2021, the Corporation completed the acquisition via a share-for-share merger of Grace Therapeutics, Inc. (“Grace”), a privately held emerging biopharmaceutical company focused on developing innovative drug delivery technologies for the treatment of rare and orphan diseases. The post-merger Corporation is focused on building a late-stage specialty pharmaceutical company specializing in rare and orphan diseases and developing and commercializing products that improve clinical outcomes using its novel drug delivery technologies. The Corporation seeks to apply new proprietary formulations to existing pharmaceutical compounds to achieve enhanced efficacy, faster onset of action, reduced side effects, more convenient delivery and increased patient compliance; all of which could result in improved patient outcomes. The active pharmaceutical ingredients chosen by the Corporation for further development may be already approved in the target indication or could be repurposed for use in new indications.
The Corporation has incurred operating losses and negative cash flows from operations in each year since its inception. The Corporation expects to incur significant expenses and continued operating losses for the foreseeable future.
In May 2023, the Corporation implemented a strategic realignment plan to enhance shareholder value that resulted in the Corporation engaging a new management team, streamlining its research and development activities and greatly reducing its workforce. Following the realignment, the Corporation is a smaller, more focused organization, based in the United States, and concentrated on its development of its lead product GTX-104. Further development of GTX-102 and GTX-101 will occur at such time when the Company is able to secure additional funding, or enters into strategic partnerships for license or sale with third parties.
On September 24, 2023, the Corporation entered into a securities purchase agreement with certain institutional and accredited investors. Gross proceeds to the Corporation from this private placement were $
The Corporation will require additional capital to fund its daily operating needs beyond that time. The Corporation does not expect to generate revenue from product sales unless and until it successfully completes drug development and obtains regulatory approval, which the Corporation expects will take several years and is subject to significant uncertainty. To date, the Corporation has financed its operations primarily through public offerings and private placements of its Common Shares, warrants and convertible debt and the proceeds from research tax credits. Until such time that the Corporation can generate significant revenue from drug product sales, if ever, it will require additional financing, which is expected to be sourced from a combination of public or private equity or debt financing or other non-dilutive sources, which may include fees, milestone payments and royalties from collaborations with third parties. Arrangements with collaborators or others may require the Corporation to relinquish certain rights related to its technologies or drug product candidates. Adequate additional financing may not be available to the Corporation on acceptable terms, or at all. The Corporation’s inability to raise capital as and when needed could have a negative impact on its financial condition and its ability to pursue its business strategy. The Corporation plans to raise additional capital in order to maintain adequate liquidity. Negative results from studies or trials, if any, or depressed prices of the Corporation’s stock could impact the Corporation’s ability to raise additional financing. Raising additional equity capital is subject to market conditions that are not within the Corporation’s control. If the Corporation is unable to raise additional funds, the Corporation may not be able to realize its assets and discharge its liabilities in the normal course of business.
The Corporation remains subject to risks similar to other development-stage companies in the biopharmaceutical industry, including compliance with government regulations, protection of proprietary technology, dependence on third-party contractors and consultants and
9
potential product liability, among others. Please refer to the risk factors included in Part 1, Item 1A of the Corporation’s Annual Report on Form 10-K for the year ended March 31, 2023, filed with the SEC on June 23, 2023 (the “Annual Report”).
Reverse stock split
On June 29, 2023, the Board of Directors of the Corporation approved an amendment to the Corporation's Articles of Incorporation to implement a reverse stock split of the Corporation's Common Shares, at a ratio of
2. Summary of significant accounting policies:
Basis of presentation
The accompanying unaudited condensed consolidated financial statements have been prepared in accordance with generally accepted accounting principles in the United States of America (“U.S. GAAP”) for interim financial information and with the instructions to Form 10-Q and Article 8 of Regulation S-X under the Securities Exchange Act of 1934. Any reference in these notes to applicable guidance is meant to refer to the authoritative U.S. GAAP as found in the Accounting Standards Codification (“ASC”) and as amended by Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
The unaudited condensed consolidated financial statements have been prepared on the same basis as the audited annual consolidated financial statements as of and for the year ended March 31, 2023, and, in the opinion of management, reflect all adjustments, consisting of normal recurring adjustments, necessary for the fair presentation of the Corporation’s consolidated financial position as of December 31, 2023, the consolidated results of its operations for the three and nine months ended December 31, 2023 and 2022, its statements of shareholders’ equity for the three and nine months ended December 31, 2023 and 2022, and its consolidated cash flows for the nine months ended December 31, 2023 and 2022.
These unaudited condensed consolidated financial statements should be read in conjunction with the Corporation’s audited consolidated financial statements and the accompanying notes for the year ended March 31, 2023 included in the Corporation’s Annual Report. The condensed consolidated balance sheet data as of March 31, 2023 presented for comparative purposes was derived from the Corporation’s audited consolidated financial statements. The results for the three and nine months ended December 31, 2023 are not necessarily indicative of the operating results to be expected for the full year or for any other subsequent interim period.
The Corporation’s significant accounting policies are disclosed in the audited consolidated financial statements for the year ended March 31, 2023 included in the Annual Report. There have been no changes to the Corporation's significant accounting policies since the date of the audited consolidated financial statements for the year ended March 31, 2023 included in the Annual Report.
Use of estimates
The preparation of these financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, income, and expenses. Actual results may differ from these estimates.
Estimates are based on management’s best knowledge of current events and actions that management may undertake in the future. Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimates are revised and in any future periods affected.
Estimates and assumptions include the measurement of stock-based compensation, derivative warrant liabilities, accruals for research and development contracts and contract organization agreements, and valuation of intangibles and goodwill. Estimates and assumptions are also involved in determining the extent to which research and development expenses qualify for research and development tax credits. The Corporation recognizes tax credits once it has reasonable assurance that they will be realized.
Recent accounting pronouncements
The Corporation has considered recent accounting pronouncements and concluded that they are either not applicable to the Corporation's business or that the effect is not expected to be material to the consolidated financial statements as a result of future adoption.
3. Fair Value Measurements
Assets and liabilities measured at fair value on a recurring basis as of December 31, 2023 are as follows:
10
|
|
Total |
|
Quoted prices in active markets (Level 1) |
|
Significant other observable inputs (Level 2) |
|
Significant unobservable inputs (Level 3) |
|
|
|
$ |
|
$ |
|
$ |
|
$ |
|
Assets |
|
|
|
|
|
|
|
|
|
Guaranteed investment certificates and term deposits |
|
|
|
|
|
||||
Guaranteed investment certificates and term deposits |
|
|
|
|
|
||||
Total assets |
|
|
|
|
|
||||
Liabilities |
|
|
|
|
|
|
|
|
|
Derivative warrant liabilities |
|
|
|
|
|
||||
Total liabilities |
|
|
|
|
|
Assets measured at fair value on a recurring basis as of March 31, 2023 are as follows:
|
|
Total |
|
Quoted prices in active markets (Level 1) |
|
Significant other observable inputs (Level 2) |
|
Significant unobservable inputs (Level 3) |
|
|
|
$ |
|
$ |
|
$ |
|
$ |
|
Assets |
|
|
|
|
|
|
|
|
|
Guaranteed investment certificate classified as a |
|
|
|
|
|
||||
Total assets |
|
|
|
|
|
There were
4. Receivables
|
|
December 31, 2023 |
|
March 31, |
|
|
$ |
|
$ |
Sales tax receivables |
|
|
||
Government assistance |
|
|
||
Interest receivable |
|
|
||
Other receivables |
|
|
||
Total receivables |
|
|
11
Government assistance is comprised of research and development investment tax credits from the Québec provincial government, which relate to quantifiable research and development expenditures under the applicable tax laws. The amounts recorded as receivables are subject to a government tax audit and the final amounts received may differ from those recorded.
5. Short-term investments
The Corporation holds various marketable securities, with maturities greater than 3 months at the time of purchase, as follows:
|
|
December 31, 2023 |
|
|
March 31, |
|
||
|
|
$ |
|
|
$ |
|
||
Term deposits issued in CAD currency earning interest at |
|
|
|
|
|
|
||
Term deposits issued in USD currency earning interest at |
|
|
|
|
|
|
||
Term deposits issued in USD currency earning interest at |
|
|
|
|
|
|
||
Total short-term investments |
|
|
|
|
|
|
||
6
|
|
December 31, 2023 |
|
|
March 31, 2023 |
|
||
|
|
$ |
|
|
$ |
|
||
Trade payables |
|
|
|
|
|
|
||
Accrued liabilities and other payables |
|
|
|
|
|
|
||
Employee salaries and benefits payable |
|
|
|
|
|
|
||
Total trade and other payables |
|
|
|
|
|
|
||
7. Leases
The Corporation has historically entered into lease arrangements for its research and development and quality control laboratory facility located in Sherbrooke, Québec. As of December 31, 2023, the Corporation had one operating lease with required future minimum payments.
Supplemental balance sheet information related to leases as of December 31, 2023 was as follows:
|
|
December 31, 2023 |
|
March 31, 2023 |
|
|
$ |
|
$ |
Operating lease right of use asset |
|
|
||
Operating lease liability, current |
|
|
||
Operating lease liability, long-term |
|
|
||
Total operating lease liability |
|
|
Supplemental lease expense related to leases is as follows:
|
|
Three months ended |
|
Nine months ended |
||||
|
|
December 31, 2023 |
|
December 31, 2022 |
|
December 31, 2023 |
|
December 31, 2022 |
|
|
$ |
|
$ |
|
$ |
|
$ |
Operating lease cost |
|
|
|
|
||||
Total lease expense |
|
|
|
|
||||
12
The following table contains a summary of the lease costs recognized under ASC 842 and other information pertaining to the Corporation’s operating lease for the nine-month period ended December 31, 2023:
Operating cash flows for operating lease |
|
$ |
|
|
Weighted-average remaining lease term (in years) |
|
|
|
|
Weighted-average discount rate |
|
|
% |
As the Corporation's lease does not provide an implicit rate, the Corporation utilized its incremental borrowing rate to discount lease payments, which reflects the fixed rate at which the Corporation could borrow on a collateralized basis the amount of the lease payments in the same currency, for a similar term, in a similar economic environment.
Future minimum lease payments under the Corporation’s operating lease as of December 31, 2023 were as follows:
|
|
December 31, 2023 |
|
|
|
|
$ |
|
|
2024 |
|
|
|
|
2025 and thereafter |
|
|
|
|
Total lease payments |
|
|
|
|
Less: interest |
|
|
|
|
Total lease liability |
|
|
|
|
8. Common shares and warrants
a. Common Shares
Authorized capital stock
Unlimited number of shares
Private Placement
On September 24, 2023, the Corporation entered into a securities purchase agreement (the “Purchase Agreement”) with certain institutional and accredited investors in connection with a private placement of the Corporation's securities (the “Offering”). Pursuant to the Purchase Agreement, the Corporation agreed to offer and sell
13
the terms of the Purchase Agreement, for each Common Share and each Pre-funded Warrant issued in the Offering, an accompanying five-eighths (0.625) of a Common Warrant was issued to the purchaser thereof. Each whole Common Warrant is exercisable for one Common Share at an exercise price of $3.003 per Common Share, is immediately exercisable, and will expire on the earlier of (i) the 60th day after the date of the acceptance by the U.S. Food and Drug Administration of a New Drug Application for the Corporation’s product candidate GTX-104 or (ii) five years from the date of issuance.
The Offering closed on September 25, 2023. The net proceeds to the Corporation from the Offering were $
At-the-Market (“ATM”) Program
On June 29, 2020, the Corporation entered into an amended and restated sales agreement (the “Sales Agreement”) with B. Riley FBR, Inc. (“B.Riley”), Oppenheimer & Co. Inc. and H.C. Wainwright & Co., LLC (collectively, the “Agents”) to amend the Corporation’s existing ATM program. Under the terms of the Sales Agreement, which had a three-year term, the Corporation could issue and sell from time to time, Common Shares having aggregate gross proceeds of up to $
During the nine months ended December 31, 2023,
b. Warrants
On May 9, 2023, warrants issued pursuant to the Corporation’s May 2018 Canadian public offering to acquire
As further discussed above, on September 25, 2023, the Corporation issued Warrants exercisable for
The Common Warrants issued as a part of the Offering are derivative warrant liabilities given the warrant indenture did not meet the fixed-for-fixed criterion and that the Common Warrants are not indexed to the Corporation’s own stock. Proceeds were allocated amongst Common Shares, Pre-funded Warrants, and Common Warrants by applying the residual method, with fair value of the Common Warrants determined using the Black-Scholes model, resulting in an initial warrant liability of $
The derivative warrant liabilities are measured at fair value at each reporting period and the reconciliation of changes in fair value is presented in the following table:
|
|
|
|
|
|
December 31, 2023 |
December 31, 2022 |
|
|
$ |
$ |
Beginning balance |
|
||
Issued during the year |
|
||
Change in fair value |
|
( |
|
Ending balance |
|
The warrant liability was determined based on the fair value of warrants at the issue date and the reporting dates using the Black-Scholes model with the following weighted-average assumptions will expire on the earlier of (i) the 60th day after the date of the acceptance by the U.S. Food and Drug Administration of a New Drug Application for the Corporation's product candidate GTX-104 or (ii) five years from the date on issuance.
14
|
|
|
|
|
|
|
September 25, 2023 |
|
December 31, 2023 |
Risk-free interest rate |
|
|
||
Share price |
|
$ |
|
$ |
Expected warrant life |
|
|
||
Dividend yield |
|
|
||
Expected volatility |
|
|
The weighted-average assumptions were prorated based on the probability of the warrant liability expiring on the 60th day after the date of the acceptance by the U.S. Food and Drug Administration of a New Drug Application for the Corporation's product candidate GTX-104 and of it expiring on five years from the date of issuance. The weighted-average fair values of the Common Warrants were determined to be $
At December 31, 2023, the Corporation had outstanding Common Warrants to purchase
9. Stock-based compensation
At December 31, 2023, the Corporation had in place a stock option plan for directors, officers, employees, and consultants of the Corporation (“Stock Option Plan”).
The Stock Option Plan provides for the granting of options to purchase Common Shares. Under the terms of the Stock Option Plan, the exercise price of the stock options granted under the Stock Option Plan may not be lower than the closing price of the Corporation’s Common Shares on the Nasdaq Capital Market at the close of such market the day preceding the grant. The maximum number of Common Shares that may be issued upon exercise of options granted under the amended Stock Option Plan shall not exceed 20% of the aggregate number of issued and outstanding shares of the Corporation as of July 28, 2022. The terms and conditions for acquiring and exercising options are set by the Corporation’s Board of Directors, subject to, among others, the following limitations: the term of the options cannot exceed
The total number of options issued to any one consultant within any twelve-month period cannot exceed
The following table summarizes information about activities within the Stock Option Plan for the nine-month period ended December 31, 2023:
|
|
Number of |
|
|
Weighted-average |
|
|
Weighted-average |
|
|||
|
|
|
|
|
$ |
|
|
$ |
|
|||
Outstanding, March 31, 2023 |
|
|
|
|
|
|
|
|
|
|||
Granted |
|
|
|
|
|
|
|
|
|
|||
Exercised |
|
|
( |
) |
|
|
|
|
|
|
||
Forfeited/Cancelled |
|
|
( |
) |
|
|
|
|
|
|
||
Outstanding, December 31, 2023 |
|
|
|
|
|
|
|
|
|
|||
Exercisable, December 31, 2023 |
|
|
|
|
|
|
|
|
|
|||
15
Forfeited and cancelled options were as a result of the Corporation's restructuring that occurred during the nine months ended December 31, 2023. On July 14, 2023, the Corporation's Board of Directors approved the grant of options to purchase
The weighted-average grant date fair value of awards for options granted during the nine months ended December 31, 2023 was $
|
|
December 31, 2023 |
|
December 31, 2022 |
|
|
Weighted-average |
|
Weighted-average |
Exercise price 1 |
|
$ |
|
$ |
Share price 1 |
|
$ |
|
$ |
Dividend |
|
|
||
Risk-free interest |
|
|
||
Estimated life (years) |
|
|
||
Expected volatility |
|
|
1
Compensation expense recognized under the Stock Option Plan is summarized as follows:
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||
|
|
December 31, 2023 |
|
|
December 31, 2022 |
|
|
December 31, 2023 |
|
|
December 31, 2022 |
|
||||
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||
Research and development expenses |
|
|
|
|
|
|
|
|
|
|
|
|
||||
General and administrative expenses |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Sales and marketing expenses |
|
|
|
|
|
|
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
|
|
|
|
|
|
||||
As of December 31, 2023, there was $
Corporation equity incentive plan
The Corporation established an equity incentive plan (the “Equity Incentive Plan”) for employees, directors, and consultants. The Equity Incentive Plan provides for the issuance of restricted share units (RSUs), performance share units, restricted shares, deferred share units and other stock-based awards, subject to restricted conditions as may be determined by the Board of Directors. There were
10. Loss per share
The Corporation has generated a net loss for all periods presented, therefore diluted loss per share is the same as basic loss per share since the inclusion of potentially dilutive securities would have had an anti-dilutive effect. All currently outstanding options and warrants could potentially be dilutive in the future.
The Corporation excluded the following potential Common Shares, presented based on amounts outstanding at each period end, from the computation of diluted net loss per share attributable to common shareholders for the periods indicated because including them would have had an anti-dilutive effect:
|
|
December 31, 2023 |
|
December 31, 2022 |
Options outstanding |
|
|
||
September 2023 Common Warrants |
|
|
||
May 2018 public offering warrants |
|
|
Basic and diluted net loss per share is calculated based upon the weighted-average number of Common Shares outstanding during the period. Common Shares underlying the Pre-funded Warrants are included in the calculation of basic and diluted earnings per share.
16
11. Financial instruments
a. Concentration of credit risk
Financial instruments that potentially subject the Corporation to a concentration of credit risk consist primarily of cash, cash equivalents, and short-term investments. Cash, cash equivalents, and short-term investments are all invested in accordance with the Corporation’s Investment Policy with the primary objective being the preservation of capital and the maintenance of liquidity, which risk is managed by dealing only with highly rated Canadian and U.S. institutions. The carrying amount of financial assets, as disclosed in the consolidated balance sheets, represents the Corporation’s credit exposure at the reporting date.
b. Foreign currency risk
The Corporation is exposed to financial risk related to the fluctuation of foreign exchange rates and the degrees of volatility of those rates. Foreign currency risk is limited to the portion of the Corporation's business transactions denominated in currencies other than the Corporation's functional currency of the U.S. dollar. Fluctuations related to foreign exchange rates could cause unforeseen fluctuations in the Corporation's operating results. The Corporation does not use derivative instruments to hedge exposure to foreign exchange risk. The fluctuation of the Canadian dollar in relation to the U.S. dollar and other foreign currencies will consequently have an impact upon the Corporation’s net loss.
c. Liquidity risk
Liquidity risk is the risk that the Corporation will encounter difficulty in meeting the obligations associated with its financial liabilities that are settled by delivering cash or another financial asset. The Corporation manages liquidity risk through the management of its capital structure and financial leverage. It also manages liquidity risk by continuously monitoring actual and projected cash flows. The Board of Directors reviews and approves the Corporation's operating budgets, and reviews material transactions outside the normal course of business. The Corporation currently does not have long-term debt nor arranged committed sources of financing and is currently using existing cash and short-term investment balances to fund operations. Refer to Note 1 – Nature of Operations.
12. Commitments and contingencies
Research and development contracts and contract research organizations agreements
The Corporation utilizes contract manufacturing organizations (“CMOs”) for the development and production of clinical materials and contract research organizations (“CROs”) to perform services related to its clinical trials. Pursuant to the agreements with these CMOs and CROs, the Corporation has either the right to terminate the agreements without penalties or under certain penalty conditions. As of December 31, 2023, the Corporation has
Raw krill oil supply contract
On October 25, 2019, the Corporation signed a supply agreement with Aker BioMarine Antarctic AS. (“AKBM”) to purchase raw krill oil product for a committed volume of commercial starting material for CaPre, one of the Corporation’s former drug candidates, for a total fixed value of $
17
Legal proceedings and disputes
In the ordinary course of business, the Corporation is at times subject to various legal proceedings and disputes. The Corporation assesses its liabilities and contingencies in connection with outstanding legal proceedings utilizing the latest information available. Where it is probable that the Corporation will incur a loss and the amount of the loss can be reasonably estimated, the Corporation records a liability in its consolidated financial statements. These legal contingencies may be adjusted to reflect any relevant developments. Where a loss is not probable or the amount of loss is not estimable, the Corporation does not accrue legal contingencies. While the outcome of legal proceedings is inherently uncertain, based on information currently available, management believes that it has established appropriate legal reserves. Any incremental liabilities arising from pending legal proceedings are not expected to have a material adverse effect on the Corporation’s financial position, results of operations, or cash flows. However, it is possible that the ultimate resolution of these matters, if unfavorable, may be material to the Corporation’s financial position, results of operations, or cash flows.
13. Restructuring Costs
On May 8, 2023, the Corporation communicated its decision to terminate a substantial amount of its workforce as part of a plan that intended to align the Corporation’s organizational and management cost structure to prioritize resources to GTX-104, thereby reducing losses to improve cash flow and extend available cash resources. The Corporation incurred $
18
Item 2. Management’s Discussion and Analysis of Financial Condition and Results of Operation
This management’s discussion and analysis (“MD&A”) is presented in order to provide the reader with an overview of the financial results and changes to our consolidated balance sheet at December 31, 2023. This MD&A also explains the material variations in our results of operations for the three and nine months ended December 31, 2023 and 2022, consolidated balance sheets as of December 31, 2023 and March 31, 2023, and cash flows for the nine months ended December 31, 2023 and 2022.
Market data, and certain industry data and forecasts included in this MD&A were obtained from internal Corporation surveys and market research conducted by third parties hired by us, publicly available information, reports of governmental agencies and industry publications, and independent third-party surveys. We have relied upon industry publications as our primary sources for third-party industry data and forecasts. Industry surveys, publications and forecasts generally state that the information they contain has been obtained from sources believed to be reliable, but that the accuracy and completeness of that information are not guaranteed. We have not independently verified any of the data from third-party sources or the underlying economic assumptions they have made. Similarly, internal surveys, industry forecasts and market research, which we believe to be reliable based upon our management’s or contracted third parties’ knowledge of our industry, have not been independently verified. Our estimates involve risks and uncertainties, including assumptions that may prove not to be accurate, and these estimates and certain industry data are subject to change based on various factors, including those discussed in this quarterly report and in our most recently filed Annual Report on Form 10-K, filed with the Securities and Exchange Commission (the “SEC”) on June 23, 2023 (the “Annual Report”). This MD&A contains forward-looking information. You should review our Special Note Regarding Forward-Looking Statements presented at the beginning of this quarterly report.
This MD&A should be read in conjunction with our unaudited condensed consolidated interim financial statements for the three and nine months ended December 31, 2023 and 2022 included elsewhere in this quarterly report. Our interim financial statements were prepared in accordance with U.S. GAAP.
All amounts appearing in this MD&A for the period-by-period discussions are in thousands of U.S. dollars, except share and per share amounts or unless otherwise indicated.
Business Overview
We are focused on developing and commercializing products for rare and orphan diseases that have the potential to improve clinical outcomes by using our novel drug delivery technologies. We seek to apply new proprietary formulations to approved and marketed pharmaceutical compounds to achieve enhanced efficacy, faster onset of action, reduced side effects, more convenient drug delivery and increased patient compliance; all of which could result in improved patient outcomes. The active pharmaceutical ingredients used in the drug candidates under development by Acasti may be already approved in a target indication or could be repurposed for use in new indications.
The existing well understood efficacy and safety profiles of these marketed compounds provides the opportunity for us to utilize the Section 505(b)(2) regulatory pathway under the Federal Food, Drug and Cosmetic Act for the development of our reformulated versions of these drugs, and therefore may provide a potentially shorter path to regulatory approval. Under Section 505(b)(2), if sufficient support of a product’s safety and efficacy either through previous U.S. Food and Drug Administration ("FDA") experience or sufficiently within the existing and accepted scientific literature, can be established, it may eliminate the need to conduct some of the pre-clinical studies and clinical trials that new drug candidates might otherwise require.
Our therapeutic pipeline consists of three unique clinical-stage and multiple pre-clinical stage assets supported by an intellectual property portfolio of more than 40 granted and pending patents in various jurisdictions worldwide. These drug candidates aim to improve clinical outcomes in the treatment of rare and orphan diseases by applying proprietary formulation and drug delivery technologies to existing pharmaceutical compounds to achieve improvements over the current standard of care, or to provide treatment for diseases with no currently approved therapies.
We believe that rare disorders represent an attractive area for drug development, and there remains an opportunity for us to utilize already approved drugs that have established safety profiles and clinical experience to potentially address significant unmet medical needs. A key advantage of pursuing therapies for rare disorders is the potential to receive orphan drug designation (“ODD”) from the FDA. Our three drug candidates have received ODD status, provided certain conditions are met at new drug application ("NDA") approval. ODD provides for seven years of marketing exclusivity in the United States post-launch, provided certain conditions are met, and the potential for faster regulatory review. ODD status can also result in tax credits of up to 50% of clinical development costs conducted in the United States upon marketing approval and a waiver of the NDA fees, which we estimate can translate into savings of approximately $3.2 million for our lead drug candidate, GTX-104. Developing drugs for rare diseases can often allow for clinical trials that are more manageably scaled and may require a smaller, more targeted commercial infrastructure.
The specific diseases targeted for drug development by us are well understood, although the patient populations suffering from such diseases may remain poorly served by available therapies or, in some cases, approved therapies do not yet exist. We aim to effectively treat debilitating symptoms that result from these underlying diseases.
19
Our lead drug candidate:
Other pipeline drug candidates:
In May 2023, we announced the strategic decision to prioritize development of GTX-104 with a goal to advance the product candidate to commercialization, while conserving resources as much as possible to complete development efficiently. We estimate that the deferral of GTX-102 and GTX-101 clinical development could be at least three years given the timeline to complete the development and potential commercial launch of GTX-104. Further development of GTX-102 and GTX-101 will occur at such time as we obtain additional funding, or enter into strategic partnerships for license or sale with third parties.
The decision to defer further development of GTX-102 and GTX-101 triggered a comprehensive impairment review of our intangible assets as of March 31, 2023. Given the extended timeline, we increased the discount rates used to value the related assets in order to recognize additional risks related to prioritizing one asset over the others, the financing for the projects given limited available resources and the need to preserve cash to advance GTX-104 as far as possible, potential competitor advances that could arise over three years, the general market depression affecting small cap development companies like us, and the prohibitively high dilution and expense of available funding in the capital markets. Increasing the discount rates significantly reduced the discounted cash flow values for each of the programs deferred. Accordingly, in the year ended March 31, 2023, we recorded impairment charges related to GTX-102 and GTX-101 of $22.7 million and $6.0 million respectively, together with further adjustments made to deferred taxes and goodwill directly related to those assets. The aggregate impairment charge was $33.5 million. We continue to believe that GTX-102 and GTX-101 may eventually provide significant value when development resumes and, if approved, commercialized successfully.
Our management team possesses significant experience in drug formulation and drug delivery research and development, clinical and pharmaceutical development and manufacturing, regulatory affairs, and business development, as well as being well-versed in late-stage drug development and commercialization. Importantly, our team is comprised of industry professionals with deep expertise and knowledge, including a world-renowned practicing neurosurgeon-scientist and respected authority in aSAH, as well as product development, chemistry, manufacturing and controls (“CMC”), planning, implementation, management, and execution of global Phase 2 and Phase 3 trials for a drug candidate for aSAH.
GTX-104 Overview
Nimodipine was granted FDA approval in 1988, and is the only approved drug that has been clinically shown to improve neurological outcomes in aSAH patients. It is only available in the United States as a generic oral capsule and as a branded oral liquid solution called NYMALIZE, which is manufactured and sold by Arbor Pharmaceuticals (acquired in September 2021 by Azurity Pharmaceuticals). Nimodipine has poor water solubility and high permeability characteristics as a result of its high lipophilicity. Additionally, orally administered nimodipine has dose-limiting side-effects such as hypotension, poor absorption and low bioavailability resulting from high first-pass metabolism, and a narrow administration window as food effects lower bioavailability significantly. Due to these issues, blood levels of orally administered nimodipine can be highly variable, making it difficult to manage blood pressure in aSAH patients. Nimodipine capsules are also difficult to administer, particularly to unconscious patients or those with impaired ability to swallow. Concomitant use with CYP3A inhibitors is contraindicated (NIMODIPINE Capsule PI).
NIMOTOP is an injectable form of nimodipine that is manufactured by Bayer Healthcare. It is approved in Europe and in other regulated markets (but not in the United States). It has limited utility for aSAH patients because of its high organic solvent content, namely 23.7% ethanol and 17% polyethylene glycol 400 (NIMOTOP SmPC).
20
Key Potential Benefits:
GTX-104 could provide a more convenient mode of administration as compared to generic nimodipine capsules or NYMALIZE. GTX-104 is administered as an intravenous infusion compared to oral administration via a nasogastric tube in unconscious patients every four hours for both nimodipine capsules and NYMALIZE. Therefore, GTX-104 could make a major contribution to patient care by potentially reducing the dosing associated nursing burden. More convenient, continuous, and consistent dosing can also reduce the risk of medication errors. In addition, as depicted in the charts below, two PK studies have shown that GTX-104 has the potential to provide improved bioavailability and show reduced inter- and intra-subject variability compared to oral nimodipine, which is hypothesized to limit the risk of hypotension and to better achieve a desired therapeutic concentration. The variability was observed higher following the capsule administration as compared to IV infusion administration (nimodipine exposure variability at steady state observed 37.5% following oral capsule administration versus 15.5%, following GTX-104 IV infusion) Because of its IV formulation, we also expect GTX-104 to reduce certain drug-drug interactions and food effects.

Despite the positive impact it has on recovery, physicians often must discontinue their patients from oral nimodipine, primarily as a result of hypotensive episodes that cannot be controlled by titrating the oral form of drug. Such discontinuation could potentially be avoided by
21
administering GTX-104, which because of its IV administration, may reduce the complexity associated with the need for careful attention to the timing of nimodipine administration at least one hour before or two hours after a meal. Also, unconscious patients will likely receive more consistent concentrations of nimodipine when delivered via the IV route as compared to oral gavage or a nasogastric tube. More consistent dosing is expected to result in a reduction of vasospasm and a better, more consistent management of hypotension. As summarized in the table below, we also anticipate reduced use of rescue therapies, such as vasopressors, and expensive hospital resources, such as the angiography suite, are possible by more effectively managing blood pressure with GTX-104. Reduced incidences of vasospasm could result in shorter length of stay and better outcomes.

About aneurysmal Subarachnoid Hemorrhage (aSAH)
aSAH is bleeding over the surface of the brain in the subarachnoid space between the brain and the skull, which contains blood vessels that supply the brain. A primary cause of such bleeding is rupture of an aneurysm. The result is a relatively uncommon type of stroke that accounts for about 5% of all strokes and has an incidence of six per 100,000 person years.
In contrast to more common types of stroke in elderly individuals, aSAH often occurs at a relatively young age, with approximately half the affected patients younger than 60 years old. Approximately 10% to 15% of aneurysmal SAH (“aSAH”) patients die before reaching the hospital, and those who survive the initial hours post hemorrhage are admitted or transferred to tertiary care centers with high risk of complications, including rebleeding and delayed cerebral ischemia (“DCI”). Systemic manifestations affecting cardiovascular, pulmonary, and renal function are common and often complicate management of DCI. Approximately 70% of aSAH patients experience death or a permanent dependence on family members, and half die within one month after the hemorrhage. Of those who survive the initial month, half remain permanently dependent on a caregiver to maintain daily living.
We estimate that approximately 50,000 individuals experience aSAH each year in the U.S. based on third-party market research, and that total addressable market for aSAH is approximately $300 million in the U.S. There are an estimated 150,000 aSAH patients each year in China and approximately 55,000 patients in the European Union based on annual inpatient admissions and the average length-of-stay.
GTX-104 Development Milestones
In September 2021, we initiated our pivotal PK bridging trial to evaluate the relative bioavailability of GTX-104 compared to currently marketed oral nimodipine capsules in approximately 50 healthy subjects. The PK trial was the next required step in our proposed 505(b)(2) regulatory pathway for GTX-104.
Final results from this pivotal PK trial were reported on May 18, 2022, and showed that the bioavailability of GTX-104 compared favorably with the oral formulation of nimodipine in all subjects, and no serious adverse events were observed for GTX-104.
All three endpoints indicated that statistically there was no difference in exposures between GTX-104 and oral nimodipine over the defined time periods for both maximum exposure and total exposure. Plasma concentrations obtained following IV administration showed significantly less variability between subjects as compared to oral administration of capsules, since IV administration is not as sensitive to some of the physiological processes that affect oral administration, such as taking the drug with and without meals, variable gastrointestinal transit time, variable drug uptake from the gastrointestinal tract into the systemic circulation, and variable hepatic blood flow and hepatic
22
first pass metabolism. Previous studies have shown these processes significantly affect the oral bioavailability of nimodipine, and therefore cause oral administration to be prone to larger inter- and intra-subject variability.
The bioavailability of oral nimodipine capsules observed was only 8% compared to 100% for GTX-104. Consequently, about one-twelfth the amount of nimodipine is delivered with GTX-104 to achieve the same blood levels as with the oral capsules.
No serious adverse events and no adverse events leading to withdrawal were reported during the trial.
Phase 3 STRIVE-ON Randomized Safety Trial for GTX-104
In April 2023, we received a Type C written meeting response and clarifying feedback from the FDA on our proposed Phase 3 safety trial for GTX-104. The FDA provided additional comments on our development plan that, pending submission of the final clinical protocol and FDA approval of same, would allow us to proceed with the initiation of a Phase 3 safety clinical trial in aSAH patients. On July 5, 2023, we announced the alignment with the U.S. Food and Drug Administration on our GTX-104 pivotal Phase 3 safety trial protocol.
The FDA concurred with the suitability of the 505(b)(2) regulatory pathway with the selected Reference Listed Drug NIMOTOP oral capsules (NDA 018869), and that our GTX-104-002 PK trial may have met the criteria for a scientific bridge.
Based on the design of our Phase 3 trial, which we have titled STRIVE-ON (Safety, Tolerability, Randomized, IV and Oral Nimodipine), the clinical trial is a prospective, open-label, randomized (1:1 ratio), parallel group trial of GTX-104 compared with oral nimodipine, in patients hospitalized for aSAH. Key trial design features include:
On October 23, 2023, we enrolled our first patient in our STRIVE-ON clinical trial. The trial is expected to take approximately 18 months to complete from the time the first patient is enrolled, and we expect this safety trial to be the final clinical step required to seek FDA approval under the 505(b)(2) regulatory pathway. Before submitting an NDA, we plan to hold a pre-NDA meeting with the FDA.
GTX-102 Overview
GTX-102 is a novel, concentrated oral-mucosal spray of betamethasone intended to improve neurological symptoms of A-T for which there are currently no FDA-approved therapies. GTX-102 is a stable, concentrated oral spray formulation comprised of the gluco-corticosteroid betamethasone that, together with other excipients can be sprayed conveniently over the tongue of the A-T patient and is rapidly absorbed.
About Ataxia Telangiectasia
A-T is a rare genetic progressive autosomal recessive neurodegenerative disorder that affects children, with the hallmark symptoms of cerebellar ataxia and other motor dysfunction, and dilated blood vessels (telangiectasia) that occur in the sclera of the eyes. A-T is caused by mutations in the ataxia telangiectasia gene, which is responsible for modulating cellular response to stress, including breaks in the double strands of DNA.
23
Children with A-T begin to experience balance and coordination problems when they begin to walk (toddler age), and ultimately become wheelchair-bound in their second decade of life. In pre-adolescence (between ages 5 and 8), patients experience oculomotor apraxia, dysarthria, and dysphagia. They also often develop compromised immune systems and are at increased risk of developing respiratory tract infections and cancer (typically lymphomas and leukemia).
A-T is diagnosed through a combination of clinical assessment (especially neurologic and oculomotor deficits), laboratory analysis, and genetic testing. There is no known treatment to slow disease progression, and treatments that are used are strictly aimed at controlling the symptoms (e.g., physical, occupational or speech therapy for neurologic issues), or conditions secondary to the disease (e.g., antibiotics for lung infections, chemotherapy for cancer, etc.). There are no FDA-approved therapeutic options currently available. Patients typically die by age 25 from complications of lung disease or cancer. According to a third-party report we commissioned, A-T affects approximately 4,300 patients per year in the United States and has a potential total addressable market of $150 million, based on the number of treatable patients in the United States.
GTX-102 - R&D and Clinical Trials to Date
We have licensed the data from the multicenter, double-blinded, randomized, placebo-controlled crossover trial from Azienda Ospedaliera Universitaria Senese, Siena, Italy, where Dr. Zannolli et. al. studied the effect of oral liquid solution of betamethasone to reduce ataxia symptoms in patients with A-T. This oral liquid solution is not marketed in the United States, and therefore is not available for clinical use; currently, betamethasone is only available in the United States as an injectable or as a topical cream. This license gives us the right to reference the trial’s data in our NDA filing. On November 12, 2015, we submitted the data from the Zannolli trial to the FDA’s Division of Neurology at a pre-Investigational New Drug (“IND”) meeting and received guidance from the agency on the regulatory requirements to seek approval.
In a multicenter, double-blind, randomized, placebo-controlled crossover trial conducted in Italy, Dr. Zannolli et al. studied the effect of an oral liquid solution of betamethasone on the reduction of ataxia symptoms in 13 children (between ages 2 to 8 years) with A-T. The primary outcome measure was the reduction in ataxia symptoms as assessed by the International Cooperative Ataxia Rating Scale (“ICARS”).
In the trial, oral liquid betamethasone reduced the ICARS total score by a median of 13 points in the intent-to-treat population and 16 points in the per-protocol population (the median percent decreases of ataxia symptoms of 28% and 31%, respectively). Adverse events in the trial were minimal, with no compulsory withdrawals and only minor side effects that did not require medical intervention. Clinical trial results in A-T patients administered oral betamethasone indicated that betamethasone significantly reduced ICARS total score relative to placebo (P = 0.01). The median ICARS change score (change in score with betamethasone minus change in score with placebo) was -13 points (95% confidence interval for the difference in medians was -19 to -5.5 points).
Based on the Zannolli data, we believe that our GTX-102 concentrated oral spray has the potential to provide clinical benefits in decreasing A-T symptoms, including assessments of posture and gait disturbance and kinetic, speech and oculomotor functions. In addition, GTX-102 may ease drug administration for patients experiencing A-T given its application of 1-3x/day of 140µL of concentrated betamethasone liquid sprayed onto the tongue using a more convenient metered dose delivery system, as these A-T patients typically have difficulty swallowing.
24
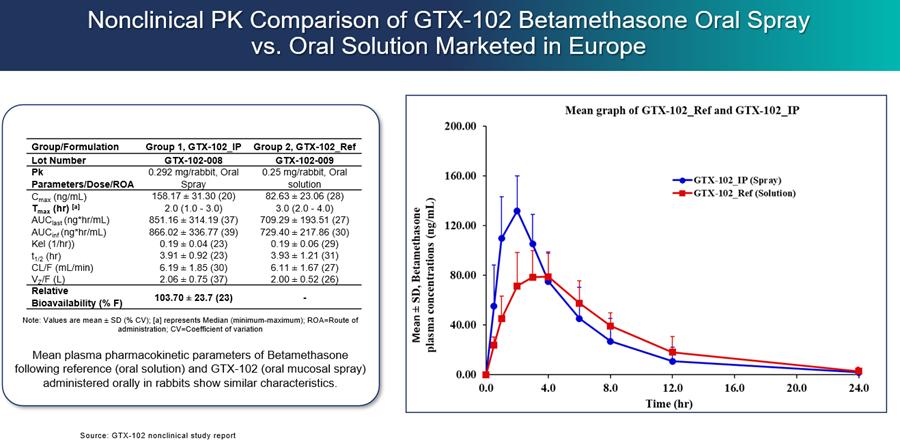
GTX-102 PK Data to Date:
GTX-102 administered as a concentrated oral spray achieves similar blood levels at only 1/70th the volume of an oral solution of betamethasone. This more convenient mode of administration will be important for A-T patients who have difficulties swallowing large volumes of liquids.
We initiated a PK bridging trial of GTX-102 as compared to the oral liquid solution of betamethasone used in the Zannolli trial and against the injectable form of betamethasone that is approved in the U.S. in the third calendar quarter of 2022. The primary objectives of the PK bridging trial were to evaluate the bioavailability, pharmacokinetics and safety of GTX-102. On December 28, 2022, we reported that the topline results of this trial met all primary outcome measures.
Results showed that GTX-102 betamethasone blood concentrations were highly predictable and consistent based on AUC (the area under the concentration time curve up to 72 hours post-dose, extrapolated to infinity) and Cmax (the maximum concentration occurring between 0 hour to 72 hours after trial drug administration), indicating good linearity and dose-proportionality. GTX-102 betamethasone blood concentrations were within the same range of exposure as IM betamethasone, based on AUC. This IM formulation will serve as a bridge for GTX-102 in the context of the proposed 505(b)(2) regulatory pathway. GTX-102 betamethasone blood concentrations were also within the same range of exposure as Oral Solution (OS), based on AUC. This OS formulation was used by Zannolli and may serve as a clinical comparator for further clinical development. Furthermore, statistically there was no significant difference (p>0.05) between GTX-102 administered at a fast rate (each spray immediately following the preceding one) vs. a slow rate (1 spray/minute), as indicated by Cmax and AUC. We believe this result is important because being able to use the fast or the slow rate of administration may provide greater flexibility for patients and caregivers. The Cmax of GTX-102 was within the same range of exposure as the OS, but the Cmax for the IM formulation was lower than both GTX-102 and the OS, as well as what has been reported previously for the IM in industry publications. It is important to note that achieving bioequivalence with the IM was not an objective of this trial, nor was it expected. Finally, of the 48 healthy adult subjects, no serious adverse events (AE) were reported, and the most frequent drug-related adverse effect was mild headache (4 cases).
The further development of GTX-102 has been deprioritized in favor of our focus on development of GTX-104. Pending additional funding for GTX-102 or the signing of a strategic partnership, we will work with our clinical experts and the FDA to determine the best final dosing regimen for GTX-102 to incorporate into our Phase 3 trial design. Based on previous discussions with the FDA, we plan to conduct a
25
confirmatory Phase 3 safety and efficacy trial in A-T patients, and plan to seek guidance from the FDA on the trial design at a Type B meeting if and when development of GTX-102 resumes. It is also possible that we may out-license or sell our GTX-102 drug candidate.
GTX-101 Overview
GTX-101 is a non-narcotic, topical bio-adhesive film-forming bupivacaine spray designed to ease the symptoms of patients suffering with postherpetic neuralgia (“PHN”). GTX-101 is administered via a metered-dose of bupivacaine spray and forms a thin bio-adhesive topical film on the surface of the patient’s skin, which enables a touch-free, non-greasy application. It also comes in convenient, portable 30 ml plastic bottles. Unlike oral gabapentin and lidocaine patches, we believe that the biphasic delivery mechanism of GTX-101 has the potential for rapid onset of action and continuous pain relief for up to eight hours. No skin sensitivity was reported in a Phase 1 trial.
About Postherpetic Neuralgia (PHN)
PHN is neuropathic pain due to damage caused by the varicella zoster virus (“VZV”). Infection with VZV causes two distinct clinical conditions. Primary VZV infection causes varicella (i.e., chickenpox), a contagious rash illness that typically occurs among young children. Secondary VZV can reactivate clinically, decades after initial infection, to cause herpes zoster (“HZ”), otherwise known as shingles. Acute HZ arises when dormant virus particles, persisting within an affected sensory ganglion from the earlier, primary infection with VZV become reactivated when cellular immunity to varicella decreases. Viral particles replicate and may spread to the dorsal root, into the dorsal horn of the spinal cord, and through peripheral sensory nerve fibers down to the level of the skin. Viral particles also may circulate in the blood. This reactivation is accompanied by inflammation of the skin, immune response, hemorrhage, and destruction of peripheral and central neurons and their fibers. Following such neural degeneration, distinct types of pathophysiological mechanisms involving both the central and peripheral nervous systems may give rise to the severe nerve pain associated with PHN.
While the rash associated with HZ typically heals within two to four weeks, the pain may persist for months or even years, and this PHN manifestation is the most common and debilitating complication of HZ. There is currently no consensus definition for PHN, but it has been suggested by the Centers for Disease Control and Prevention (“CDC”) that PHN is best defined as pain lasting at least three months after resolution of the rash.
PHN is associated with significant loss of function and reduced quality of life, particularly in the elderly. It has a detrimental effect on all aspects of a patient's quality of life. The nature of PHN pain varies from mild to severe, constant, intermittent, or triggered by trivial stimuli. Approximately half of patients with PHN describe their pain as “horrible” or “excruciating,” ranging in duration from a few minutes to constant on a daily or almost daily basis. The pain can disrupt sleep, mood, work, and activities of daily living, adversely impacting the
26
quality of life and leading to social withdrawal and depression. PHN is the number-one cause of intractable, debilitating pain in the elderly, and has been cited as the leading cause of suicide in chronic pain patients over the age of 70.
Current treatment of PHN most often consists of oral gabapentin (first line) and prescription lidocaine patches or antidepressants (second line), and refractory cases may be prescribed opioids to address persistent pain. Gabapentin and opioid abuse have continued to proliferate, and lidocaine patches are suboptimal for many reasons. An independent third-party market research firm we commissioned interviewed more than 250 physicians who regularly treat PHN patients and found that approximately 40% of patients using lidocaine patches experience insufficient pain relief. Lidocaine patches are difficult to use, fall off, and look unsightly with possible skin sensitivity and irritation. Additionally, lidocaine patches can only be used for 12 hours and then need to be removed for 12 hours before being reapplied. Prescription lidocaine patches are only approved for PHN, and the market is currently made up of both branded and generic offerings. It is estimated that PHN affects approximately 120,000 patients per year in the United States. According to a third-party report we commissioned, the total addressable market for GTX-101 could be as large as $2.5 billion, consisting of approximately $200 million for PHN pain and $2.3 billion for non-PHN pain indications.
GTX-101 R&D History and Clinical Trials Completed to Date
To date, we have conducted four Phase I trials in healthy volunteers to assess the PK, safety and tolerability of GTX-101 and to determine the plasma levels of bupivacaine HCl administered as a single dose in various concentrations between 30 mg (three sprays) and 2100 mg (twenty sprays).
These trials confirmed that bupivacaine delivered as a topical spray (GTX-101) is well absorbed through the skin, as demonstrated in the graph below, while very little is absorbed systemically.
In all four trials, the administration of GTX-101 to healthy volunteers was safe and well tolerated. In addition, no evidence of skin irritation was observed at the application site following the spray administrations. The data below is from two separate trials of GTX-101 and the Lidoderm patch superimposed on each other.
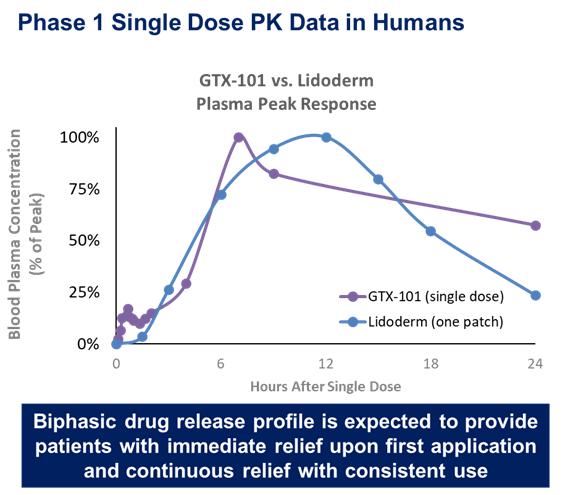
GTX-101 recent activities:
We believe that the PHN pain market will continue to grow, and non-opioid products like GTX-101 that can relieve PHN pain more quickly and in a sustained manner by means of a more efficient delivery system, will be an attractive therapy option for patients and physicians. GTX-101 is administered by spraying our proprietary bupivacaine formulation over the affected area, which we believe has the potential to provide several advantages over currently marketed products such as the lidocaine patch, including faster onset of action, sustained pain relief, possibly lower dosing requirements and improved dosing convenience, all which could lead to increased patient satisfaction and compliance.
The data from the single dose Phase 1 clinical trial for GTX-101 was submitted to the FDA’s Division of Anesthesiology and feedback was received at a pre-IND meeting on April 18, 2018, that informed the design of pre-clinical toxicology studies and a clinical and regulatory pathway to approval under section 505(b)(2). We completed a minipig skin sensitivity study in the second calendar quarter of 2022, and we
27
initiated a single dose PK trial in healthy human volunteers in July 2022. Topline results from this single dose PK trial were reported on December 23, 2022 and the results met all primary outcome measures.
The median Tmax (the time of maximum concentration between 0 hour and 240 hours after study drug administration) of bupivacaine in plasma following GTX-101 single-dose topical applications ranged between 18 to 24 hours depending on dose, while the median Tmax following the subcutaneous injection of 10 mg of bupivacaine was only 23 minutes. This result suggests that bupivacaine delivered by GTX-101 remains in the skin for a long period of time, potentially inducing prolonged analgesic effect in the sprayed area. The exposure to bupivacaine based on Cmax (the maximum concentration occurring at Tmax between 0 hour and 240 hours after study drug administration) and AUC (the area under the concentration time curve, extrapolated to infinity) following GTX-101 topical application as a single-dose increased with increasing dose.
The systemic exposure to bupivacaine following a 200mg dose of GTX-101 was approximately 29-fold less than a single subcutaneous dose of 10mg of bupivacaine based on Cmax and approximately 6-fold less than a single subcutaneous dose of 10mg of bupivacaine based on AUC. We predict these lower blood levels will correspond to an increased safety margin for GTX-101 with regards to toxicity risk. Mean half-life (T half) following GTX-101 single-dose topical applications ranged between 24 to 37 hours depending on dose, suggesting a slow elimination and potentially long duration of effect, while mean Tmax following the subcutaneous injection of 10 mg of bupivacaine was only 8 hours.
There were only two adverse events judged as related to the study drug by the investigator for each of GTX-101 and the bupivacaine subcutaneous injection. Following GTX-101 topical application: headache (1 event = 3%) and numbness (1 event = 3%) at the sprayed area following bupivacaine subcutaneous injection: dizziness (1 event = 8%) and nausea (1 event = 8%).
The further development of GTX-101 has been deprioritized in favor of our focus on development of GTX-104. Pending additional funding for GTX-101 or the signing of a strategic partnership, we plan to follow this successful PK trial with a multiple ascending dose trial in 2023. Results from these non-clinical studies and clinical trials are required before the initiation of our Phase 2 program in PHN patients. It is also possible that we may out-license or sell our GTX-101 drug candidate.
28
Overall Commercialization Strategy
We have worldwide commercialization rights for all our pipeline drug candidates and plan to maximize the value of each asset. Currently, we have prioritized the development of GTX-104 and de-emphasized the development of GTX-102 and GTX-101. If we receive regulatory approval for GTX-104 in the US, we may look to out-license commercialization or consider self-commercialization including outsourcing sales to ensure efficient commercial management and maximize market penetration and financial returns. We may seek commercial partnerships to fully exploit the market potential of GTX-104 in territories outside the US. It is possible that we may out-license or sell GTX-102 and/or GTX-101 for the US and/or global markets.
Recent Developments
Change in Certifying Accountant
On December 11, 2023, the Audit Committee of the Corporation's Board of Directors (the "Board") recommended to the Board and the Board approved the dismissal of Ernst & Young LLP (Canada) ("E&Y") as the Corporation’s independent registered public accounting firm. The report of E&Y on the consolidated financial statements of the Corporation as of and for the fiscal year ended March 31, 2023 did not contain any adverse opinion or a disclaimer of opinion, nor were they qualified or modified as to uncertainty, audit scope or accounting principles. On December 11, 2023, in connection with the Corporation’s dismissal of E&Y, the Audit Committee recommended to the Board and the Board approved the engagement of KPMG LLP (U.S.) ("KPMG") as its new independent registered public accounting firm to audit the Corporation’s consolidated financial statements for the fiscal year ending March 31, 2024. The decision to engage KPMG was recommended by the Audit Committee, and approved by the Board, after taking into account KPMG’s location in the United States, the results of a competitive review process and other business factors.
Dosing of First Patient
On October 23, 2023, we enrolled our first patient in our STRIVE-ON Phase 3 clinical trial. The trial is expected to take approximately 18 months to complete from the time the first patient is enrolled, and we expect this safety trial to be the final clinical step required to seek FDA approval under the 505(b)(2) regulatory pathway.
September 2023 Private Placement Offering
On September 24, 2023, we entered into a securities purchase agreement (the “Purchase Agreement”) with certain institutional and accredited investors in connection with a private placement offering of our securities (the “Offering”). Pursuant to the Purchase Agreement, we agreed to offer and sell 1,951,371 Class A common shares, no par value per share (the “Common Shares”), at a purchase price of $1.848 per Common Share and pre-funded warrants (the “Pre-funded Warrants”) to purchase up to 2,106,853 Common Shares at a purchase price equal to the purchase price per Common Share less $0.0001. Pursuant to the Purchase Agreement, we also issued to such institutional and accredited investors common warrants (the “Common Warrants” and, together with the Pre-funded Warrants, the “Warrants”) to purchase Common Shares, exercisable for an aggregate of 2,536,391 Common Shares. Under the terms of the Purchase Agreement, for each Common Share and each Pre-funded Warrant issued in the Offering, an accompanying five-eighths (0.625) of a Common Warrant was issued to the purchaser thereof.
The Offering closed on September 25, 2023. Shore Pharma LLC, an entity controlled by Vimal Kavuru, the Chair of our Board of Directors, and SS Pharma LLC, the beneficial owner of 5.5% of our Common Shares outstanding prior to the Offering, each a related party of ours, participated in the Offering. The net proceeds to us from the Offering were approximately $7.3 million, after deducting fees and expenses.
Pursuant to the terms of the Purchase Agreement, we agreed to register for resale the Common Shares sold in the Offering and the Warrant Shares. On October 6, 2023, we filed a resale Registration Statement on Form S-3 with the SEC, registering the Common Shares sold in the Offering and the Warrant Shares for resale. The resale Registration Statement on Form S-3 was declared effective on October 16, 2023.
Announcement of compliance with the Nasdaq minimum bid price requirement
On July 24, 2023, we received written notice (the “Notification Letter”) from The Nasdaq Stock Market LLC notifying us that we had regained compliance with the minimum bid price requirement set forth in Nasdaq Listing Rule 5550(a)(2) for continued listing on the Nasdaq Capital Market. The Notification Letter was sent following the implementation of a 1-for-6 reverse split of our Common Shares (the “Reverse Stock Split”), which became effective on July 10, 2023.
Reverse stock split
On June 29, 2023, our Board of Directors approved an amendment to our Articles of Incorporation to implement the Reverse Stock Split. On July 4, 2023, we filed Articles of Amendment to our Articles of Incorporation with the Registraire des entreprises du Québec, to implement the Reverse Stock Split. All applicable references in this MD&A to number of Common Shares, warrants and options, price per share and weighted-average number of shares outstanding prior to the Reverse Stock Split have been adjusted to reflect the Reverse Stock Split, which was made effective on July 10, 2023.
29
Announcement of successful submission of pivotal GTX-104 Phase 3 safety study protocol with FDA and implementation of strategic realignment plan
On May 8, 2023, we announced the successful submission to the FDA of GTX-104's full protocol of our pivotal Phase 3 safety trial and implementation of a strategic realignment plan to maximize shareholder value.
Key strategies implemented were:
In connection with the transformation of our operating model, we appointed the following industry experts to our senior management team:
Following the realignment, the Corporation is a smaller, more focused organization, based in the United States, and concentrated on its development of its lead product GTX-104.
Basis of Presentation of the Financial Statements
Our condensed consolidated interim financial statements, which include the accounts of our wholly owned subsidiaries, Acasti Pharma U.S., and Acasti Innovations AG, have been prepared in accordance with U.S. GAAP and the rules and regulations of the SEC related to quarterly reports filed on Form 10-Q. All intercompany transactions and balances are eliminated on consolidation.
Our assets as of December 31, 2023, include cash, cash equivalents, and short-term investments totaling $25.1 million and intangible assets and goodwill totaling $49.3 million. Our current liabilities total $1.8 million as of December 31, 2023 and are comprised primarily of amounts due to or accrued for creditors. The Corporation believes its cash runway will be sufficient to fund the Corporation’s operations into the second calendar quarter of 2026.
30
Results of Operations for the Three and Nine months ended December 31, 2023 and 2022
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||||||||||
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
||||||
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Operating expenses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Research and development expenses, net of government assistance |
|
|
1,443 |
|
|
|
2,450 |
|
|
|
(1,007 |
) |
|
|
2,998 |
|
|
|
8,332 |
|
|
|
(5,334 |
) |
General and administrative expenses |
|
|
1,570 |
|
|
|
1,589 |
|
|
|
(19 |
) |
|
|
4,922 |
|
|
|
5,187 |
|
|
|
(265 |
) |
Sales and marketing expenses |
|
|
30 |
|
|
|
206 |
|
|
|
(176 |
) |
|
|
184 |
|
|
|
563 |
|
|
|
(379 |
) |
Restructuring costs |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,485 |
|
|
|
— |
|
|
|
1,485 |
|
Loss from operating activities |
|
|
(3,043 |
) |
|
|
(4,245 |
) |
|
|
(1,202 |
) |
|
|
(9,589 |
) |
|
|
(14,082 |
) |
|
|
(4,493 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Foreign exchange gain (loss) |
|
|
3 |
|
|
|
15 |
|
|
|
(12 |
) |
|
|
(2 |
) |
|
|
(75 |
) |
|
|
73 |
|
Change in fair value of warrant liabilities |
|
|
125 |
|
|
|
— |
|
|
|
125 |
|
|
|
(1,701 |
) |
|
|
10 |
|
|
|
(1,711 |
) |
Interest income and other expense |
|
|
316 |
|
|
|
67 |
|
|
|
249 |
|
|
|
662 |
|
|
|
134 |
|
|
|
528 |
|
Income tax recovery |
|
|
208 |
|
|
|
274 |
|
|
|
(66 |
) |
|
|
943 |
|
|
|
671 |
|
|
|
272 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Net loss |
|
|
(2,391 |
) |
|
|
(3,889 |
) |
|
|
(1,498 |
) |
|
|
(9,687 |
) |
|
|
(13,342 |
) |
|
|
(3,655 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Net loss
The net loss of $2,391, or $0.21 per share, for the three months ended December 31, 2023 decreased by $1,498 from the net loss of $3,889, or $0.52 per share, for the three months ended December 31, 2022.
The net loss of $9,687, or $1.09 per share, for the nine months ended December 31, 2023 decreased by $3,655 from the net loss of $13,342, or $1.80 per share, for the nine months ended December 31, 2022.
Research and development expenses, net of government assistance
Research and development expenses consist primarily of:
We record research and development expenses as incurred.
Our research and development during the three and nine months ended December 31, 2023, was focused primarily on our clinical development programs for our GTX-104 drug candidate. Research and development expenses during the three and nine months ended December 31, 2022 were focused primarily on our clinical development programs GTX-104, GTX-102, and GTX-101 drug candidates.
The following table summarizes our research and development expenses for the periods presented:
31
Research and development expenses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||||||||||
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
||||||
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||||
Total third-party research and development expenses 1 |
|
|
1,219 |
|
|
|
1,898 |
|
|
|
(679 |
) |
|
|
2,196 |
|
|
|
6,471 |
|
|
|
(4,275 |
) |
Government grants & tax credits |
|
|
— |
|
|
|
(115 |
) |
|
|
115 |
|
|
|
55 |
|
|
|
(196 |
) |
|
|
251 |
|
Salaries and benefits |
|
|
163 |
|
|
|
522 |
|
|
|
(359 |
) |
|
|
597 |
|
|
|
1,483 |
|
|
|
(886 |
) |
Research and development expense before stock-based compensation and depreciation |
|
|
1,382 |
|
|
|
2,305 |
|
|
|
(923 |
) |
|
|
2,848 |
|
|
|
7,758 |
|
|
|
(4,910 |
) |
Stock-based compensation |
|
|
61 |
|
|
|
139 |
|
|
|
(78 |
) |
|
|
145 |
|
|
|
481 |
|
|
|
(336 |
) |
Depreciation and write-off of equipment |
|
|
— |
|
|
|
6 |
|
|
|
(6 |
) |
|
|
5 |
|
|
|
93 |
|
|
|
(88 |
) |
Total |
|
|
1,443 |
|
|
|
2,450 |
|
|
|
(1,007 |
) |
|
|
2,998 |
|
|
|
8,332 |
|
|
|
(5,334 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
1 Total third-party research and development expenses are calculated before salaries and benefits, depreciation, write-off of equipment and stock-based compensation. Because there is no standard method endorsed by U.S. GAAP, the results may not be comparable to similar measurements presented by other public companies.
Total third-party research and development expenses for the three and nine months ended December 31, 2023, were $1,219 and $2,196, respectively, compared to $1,898 and $6,471 for the three and nine months ended December 31, 2022, respectively. This decrease of $679 and $4,275 was primarily due to the restructuring to align our organizational and management cost structure to prioritize resources to GTX-104, thereby reducing losses to improve cash flow and extend available cash resources. Our clinical development programs for GTX-102, and GTX-101 were de-prioritized in the current year period compared to the prior year period.
Government grants and tax credits of $0 and $55 for the three and nine months ended December 31, 2023, respectively, increased by $115 and $251 compared to $(115) and $(196) for the three and nine months ended December 31, 2022, respectively. The changes within government grants and tax credits were due to adjustments of provisions regarding the estimated realizability of credits receivable after assessments and correspondence from tax authorities.
Salaries and benefits of $163 and $597 for the three and nine months ended December 31, 2023, respectively, decreased by $359 and $886 compared to $522 and $1,483 for the three and nine months ended December 31, 2022, respectively. The decrease was primarily due to a reduction in research and development headcount due to the restructuring.
32
General and administrative expenses
General and administrative expenses consist primarily of salaries and related benefits, including stock-based compensation, related to our executive, finance, legal, and support functions, including professional fees for auditing, tax, consulting, rent and utilities and insurance.
General and administrative expenses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||||||||||
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
||||||
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||||
Salaries and benefits |
|
|
237 |
|
|
|
490 |
|
|
|
(253 |
) |
|
|
808 |
|
|
|
1,494 |
|
|
|
(686 |
) |
Professional fees |
|
|
764 |
|
|
|
406 |
|
|
|
358 |
|
|
|
2,573 |
|
|
|
1,443 |
|
|
|
1,130 |
|
Other |
|
|
304 |
|
|
|
393 |
|
|
|
(89 |
) |
|
|
1,013 |
|
|
|
1,266 |
|
|
|
(253 |
) |
General and administrative expense before stock-based compensation and depreciation 1 |
|
|
1,305 |
|
|
|
1,289 |
|
|
|
16 |
|
|
|
4,394 |
|
|
|
4,203 |
|
|
|
191 |
|
Stock-based compensation |
|
|
265 |
|
|
|
280 |
|
|
|
(15 |
) |
|
|
523 |
|
|
|
930 |
|
|
|
(407 |
) |
Depreciation |
|
|
— |
|
|
|
20 |
|
|
|
(20 |
) |
|
|
5 |
|
|
|
54 |
|
|
|
(49 |
) |
Total |
|
|
1,570 |
|
|
|
1,589 |
|
|
|
(19 |
) |
|
|
4,922 |
|
|
|
5,187 |
|
|
|
(265 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
1 General and administrative sub-total expenses are calculated before stock-based compensation and depreciation. Because there is no standard method endorsed by U.S. GAAP, the results may not be comparable to similar measurements presented by other public companies.
General and administrative expenses were $1,305 and $4,394 before stock-based compensation and depreciation expense for the three and nine months ended December 31, 2023, respectively, an increase of $16 and $191 from $1,289 and $4,203 for the three and nine months ended December 31, 2022, respectively. The increase was primarily a result of increased legal, tax, accounting and other professional fees related to the restructuring offset by decreased salaries and benefits due to a reduction in general and administrative headcount due to our restructuring and reorganization of our management structure. Stock-based compensation of $265 and $523 for the three and nine months ended December 31, 2023, respectively, decreased by $15 and $407 compared to $280 and $930 for the three and nine months ended December 31, 2022, respectively. The decrease was primarily due to a reduction in general and administrative headcount as a result of our restructuring.
Sales and marketing expenses
Sales and marketing expenses consist primarily of salaries and benefits, including stock-based compensation, related to our commercial functions.
Sales and marketing expenses |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
|
|
Three months ended |
|
|
Nine months ended |
|
||||||||||||||||||
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
|
December 31, |
|
|
December 31, |
|
|
Increase (Decrease) |
|
||||||
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
|
$ |
|
||||||
Salaries and benefits |
|
|
— |
|
|
|
109 |
|
|
|
(109 |
) |
|
|
15 |
|
|
|
390 |
|
|
|
(375 |
) |
Professional fees |
|
|
— |
|
|
|
1 |
|
|
|
(1 |
) |
|
|
20 |
|
|
|
10 |
|
|
|
10 |
|
Other |
|
|
30 |
|
|
|
72 |
|
|
|
(42 |
) |
|
|
133 |
|
|
|
85 |
|
|
|
48 |
|
Sales and Marketing expenses before stock-based compensation 1 |
|
|
30 |
|
|
|
182 |
|
|
|
(152 |
) |
|
|
168 |
|
|
|
485 |
|
|
|
(317 |
) |
Stock-based compensation |
|
|
— |
|
|
|
24 |
|
|
|
(24 |
) |
|
|
16 |
|
|
|
78 |
|
|
|
(62 |
) |
Total |
|
|
30 |
|
|
|
206 |
|
|
|
(176 |
) |
|
|
184 |
|
|
|
563 |
|
|
|
(379 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
1 Sales and marketing sub-total expenses are calculated before stock-based compensation. Because there is no standard method endorsed by U.S. GAAP, the results may not be comparable to similar measurements presented by other public companies.
Sales and marketing expenses before stock-based compensation expense were $30 and $168 for the three and nine months ended December 31, 2023, respectively, compared to $182 and $485 for the three and nine months ended December 31, 2022, respectively. The decrease of $152 and $317, was primarily due to the reduction of headcount due to our restructuring and reorganization of our management structure.
33
Stock-based compensation of nil and $16 for the three and nine months ended December 31, 2023, decreased by $24 and $62, respectively, compared to $24 and $78 for the three and nine months ended December 31, 2022, respectively. The decrease is primarily due to a reduction in sales and marketing headcount due to our restructuring.
Restructuring Costs
On May 8, 2023, we announced our decision to terminate a substantial amount of our workforce as part of a plan intended to align our organizational and management cost structure to prioritize resources to GTX-104, thereby reducing losses to improve cash flow and extend available cash resources. We incurred $1,485 of related costs primarily consisting of employee severance costs.
Change in fair value of warrant liabilities
The fair value of warrant liabilities for the three and nine months ended December 31, 2023 decreased by $125 and increased by $1,701, respectively, mainly attributable to changes in our stock price.
Interest income and other expense
Interest income was $316 and $662 for the three and nine months ended December 31, 2023, respectively, compared to $67 and $134 for the three and nine months ended December 31, 2022, respectively. The increase in our interest income was due to higher interest rates earned on average balances of cash, cash equivalents, and short-term investments.
Liquidity and Capital Resources
Cash flows and financial condition for the nine months ended December 31, 2023 and 2022
Summary
We do not expect to generate revenue from product sales unless and until we successfully complete drug development and obtain regulatory approval, which we expect will take several years and is subject to significant uncertainty. To date, we have financed our operations primarily through public offerings and private placements of our Common Shares, warrants and convertible debt and with the proceeds from research tax credits. Until such time that we can generate significant revenue from drug product sales, if ever, we will require additional financing, which we expect to be sourced from a combination of public or private equity offerings or debt financings or other non-dilutive sources, which may include fees, milestone payments and royalties from collaborations with third parties.
As of December 31, 2023, cash, cash equivalents, and short-term investments were $25,114, a decrease of $2,776 for the nine months ended, compared to cash, cash equivalents, and short-term investments of $27,890 at March 31, 2023 primarily due to operating activities, and funding the restructuring expenses, offset in part by the proceeds of our September 2023 Offering.
Net cash used in operating activities
Net cash used in operating activities for the nine months ended December 31, 2023 was $10,245, compared to $12,587 for the nine months ended December 31, 2022, a decrease of $2,342. Cash used in operating activities during the nine months ended December 31, 2023 primarily related to our net loss of $9,687, adjusted for non-cash items such as stock-based compensation of $684, change in fair value of warrant liabilities of $1,701, income tax recovery of $(943) and changes in our operating assets and liabilities of $(1,984). Net cash used in operating activities for the nine months ended December 31, 2022, was $12,587. Cash used in operating activities during 2022 primarily related to our net loss of $13,342, adjusted for non-cash items such as stock-based compensation of $1,489, income tax recovery of $671 and changes in our operating assets and liabilities of $(172).
Net cash used in investing activities
Net cash used in investing activities for the nine months ended December 31, 2023, was from our purchase of short-term investments of $6,554 offset by proceeds of $110 related to our sale of equipment. For the nine months ended December 31, 2022, our investing activities generated cash of $8,161, which was $13,185 of proceeds from maturity of short-term investments offset by our purchase of short-term investments of $5,015.
Net cash provided by financing activities
Net cash provided by financing activities for the nine months ended December 31, 2023, was $7,359 due to the net proceeds from our September 2023 Offering compared to cash provided of $304 during the nine months ended December 31, 2022 from our ATM program.
Private Placement
34
On September 24, 2023, we entered into the Purchase Agreement with certain institutional and accredited investors in connection with the Offering. Pursuant to the Purchase Agreement, we agreed to offer and sell 1,951,371 Common Shares, at a purchase price of $1.848 per Common Share and Pre-funded Warrants to purchase up to 2,106,853 Common Shares at a purchase price equal to the purchase price per Common Share less $0.0001. Each Pre-funded Warrant is exercisable for one Common Share at an exercise price of $0.0001 per Common Share, is immediately exercisable, and will expire once exercised in full. Pursuant to the Purchase Agreement, we also issued to such institutional and accredited investors Common Warrants to purchase Common Shares, exercisable for an aggregate of 2,536,391 Common Shares. Under the terms of the Purchase Agreement, for each Common Share and each Pre-funded Warrant issued in the Offering, an accompanying five-eighths (0.625) of a Common Warrant was issued to the purchaser thereof. Each whole Common Warrant is exercisable for one Common Share at an exercise price of $3.003 per Common Share, is immediately exercisable, and will expire on the earlier of (i) the 60th day after the date of the acceptance by the U.S. Food and Drug Administration of a New Drug Application for the Corporation’s product candidate GTX-104 or (ii) five years from the date of issuance.
The Offering closed on September 25, 2023. The net proceeds to us from the Offering were approximately $7.3 million, after deducting fees and expenses.
At-the-Market (“ATM”) program
On June 29, 2020, we entered into an amended and restated sales agreement (the “Sales Agreement”) with B. Riley, Oppenheimer & Co. Inc. and H.C. Wainwright & Co., LLC (collectively, the “Agents”). Under the terms of the Sales Agreement, which had a three-year term, we could issue and sell from time to time, Common Shares having an aggregate offering price of up to $75 million through the Agents. Subject to the terms and conditions of the Sales Agreement, the Agents would use their commercially reasonable efforts to sell the Common Shares from time to time, based upon our instructions. We had no obligation to sell any of the Common Shares and could, at any time, suspend sales under the Sales Agreement. We and the Agents could terminate the Sales Agreement in accordance with its terms. Under the terms of the Sales Agreement, we provided the Agents with customary indemnification rights and the Agents were entitled to compensation at a commission rate equal to 3.0% of the gross proceeds from each sale of the Common Shares. The Sales Agreement expired pursuant to its terms on June 29, 2023. We intend to examine our financing strategies on a go-forward basis and may consider entering into a new ATM program in the future.
During the nine months ended December 31, 2023, no Common Shares were sold under the ATM program. During the nine months ended December 31, 2022, 54,108 Common Shares were sold for total net proceeds of $304 with commissions, legal expenses and costs related to the share sale amounting to $10. The Common Shares were sold at the prevailing market prices, which resulted in an average price of approximately $5.70 per share.
35
Treasury Operations
Our treasury policy is to invest cash that is not required immediately into instruments with an investment strategy based on capital preservation. Cash equivalents and short-term investments are primarily in guaranteed investment certificates, term deposits and high-interest savings accounts, which are issued and held with Canadian chartered banks, highly rated promissory notes issued by government bodies and commercial paper. We hold cash denominated in both U.S. and Canadian dollars.
Contractual Obligations and Commitments
Our contractual obligations and commitments include trade payables, operating lease obligations, CMO and CRO agreements, and the raw krill oil supply agreement, as described below.
Research and development contracts and contract research organizations agreements:
We utilize CMOs, for the development and production of clinical materials and CROs to perform services related to our clinical trials. Pursuant to the agreements with CMOs and CROs, we have either the right to terminate the agreements without penalties or under certain penalty conditions. As of December 31, 2023, we have no commitments from CMOs and $7,670 of commitments for the next twelve months to CROs.
Raw krill oil supply contract
On October 25, 2019, we signed a supply agreement with Aker BioMarine Antarctic AS. (“AKBM”) to purchase raw krill oil product for a committed volume of commercial starting material for CaPre, one of our former drug candidates, for a total fixed value of $3.1 million based on the value of krill oil at that time. As of March 31, 2022, the remaining balance of commitment amounted to $2.8 million. During the second calendar quarter of 2022, AKBM informed us that AKBM believed it had satisfied the terms of the supply agreement as to their obligation to deliver the remaining balance of raw krill oil product, and that we were therefore required to accept the remaining product commitment. We disagreed with AKBM’s position and believed that AKBM was not entitled to further payment under the supply agreement. Accordingly, no liability was recorded by us. The dispute remained unresolved as of both March 31, 2023 and 2022. On October 18, 2023, we entered into an agreement with AKBM to settle any and all potential claims regarding amounts due under the supply agreement (“Settlement Agreement”). Pursuant to the terms of the Settlement Agreement, in exchange for a release and waiver of claims arising out of the supply agreement by AKBM and any of AKBM’s affiliates, we agreed to the following: (a) AKBM retained ownership of all raw krill oil product, including amounts previously delivered to us, (b) AKBM acquired and took ownership of all production equipment related to the production of CaPre, (c) AKBM acquired and took ownership of all data from research, clinical trials and pre-clinical studies with respect to CaPre, and (d) AKBM acquired and took ownership over all rights, title and interest in and to all intellectual property rights, including all patents and trademarks, related to CaPre owned by us. Pursuant to the terms of the Settlement Agreement, AKBM acknowledged that the CaPre assets were transferred on an “as is” basis, and in connection therewith we disclaimed all representations and warranties in connection with the CaPre assets, including any representations with respect to performance or sufficiency. The value of the raw krill oil previously delivered to us, the production equipment, and the intellectual property rights related to CaPre were fully impaired in prior reporting periods and had a carrying value of nil as of March 31, 2023. As of December 31, 2023, no liability was recorded.
Contingencies
We evaluate contingencies on an ongoing basis and establish loss provisions for matters in which losses are probable and the amount of the loss can be reasonably estimated.
36
Use of Estimates and Measurement of Uncertainty
The preparation of these consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, income, and expenses. Actual results may differ from these estimates.
Estimates are based on management’s best knowledge of current events and actions that management may undertake in the future. Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimates are revised and in any future periods affected.
Estimates and assumptions include the measurement of stock-based compensation, derivative warrant liabilities, accruals for research and development contracts and contract organization agreements, and valuation of intangibles and goodwill. Estimates and assumptions are also involved in determining which research and development expenses qualify for research and development tax credits and in what amounts. The Corporation recognizes the tax credits once it has reasonable assurance that they will be realized.
Critical Accounting Policies
During the nine months ended December 31, 2023, there were no material changes to our critical accounting policies from those described in our Annual Report for the year ended March 31, 2023.
Recent Accounting Pronouncements
We have considered recent accounting pronouncements and concluded that they are either not applicable to our business or that the effect is not expected to be material to our consolidated financial statements as a result of future adoption.
Item 3. Quantitative and Qualitative Disclosures About Market Risk
A smaller reporting company is not required to provide the information required by this Item.
Item 4. Controls and Procedures
Disclosure Controls and Procedures
As of the end of the period covered by this quarterly report, our management, with the participation of our Chief Executive Officer and Principal Financial Officer, has performed an evaluation of the effectiveness of our disclosure controls and procedures within the meaning of Rules 13a-15(e) and 15d-15(e) of the Exchange Act. Based upon this evaluation, our management has concluded that, as of December 31, 2023, our existing disclosure controls and procedures were effective. It should be noted that while our Chief Executive Officer and Principal Financial Officer believe that our disclosure controls and procedures provide a reasonable level of assurance that they are effective, they do not expect the disclosure controls and procedures to be capable of preventing all errors and fraud. A control system, no matter how well conceived or operated, can provide only reasonable, but not absolute, assurance that the objectives of the control system are met.
Changes in Internal Control over Financial Reporting
No changes were made to our internal controls over financial reporting that occurred during the quarter ended December 31, 2023 that have materially affected, or are reasonably likely to materially affect, our internal controls over financial reporting.
PART II. OTHER INFORMATION
Item 1. Legal Proceedings
In the ordinary course of business, we are at times subject to various legal proceedings and disputes. We assess our liabilities and contingencies in connection with outstanding legal proceedings utilizing the latest information available. Where it is probable that we will incur a loss and the amount of the loss can be reasonably estimated, we record a liability in our consolidated financial statements. These legal reserves may be increased or decreased to reflect any relevant developments on a quarterly basis. Where a loss is not probable or the amount of loss is not estimable, we do not accrue legal reserves. While the outcome of legal proceedings is inherently uncertain, based on information currently available and available insurance coverage, our management believes that it has established appropriate legal reserves. Any incremental liabilities arising from pending legal proceedings are not expected to have a material adverse effect on our financial position, results of operations, or cash flows. However, it is possible that the ultimate resolution of these matters, if unfavorable, may be material to our financial position, results of operations, or cash flows. We are not currently a party to any legal proceedings that, in the opinion of management, are likely to have a material adverse effect on our business.
37
Item 1A. Risk Factors
There have been no material changes from the risk factors disclosed in our Annual Report.
Item 2. Unregistered Sales of Equity Securities and Use of Proceeds
None.
Item 3. Defaults upon Senior Securities
None.
Item 4. Mine Safety Disclosures
Not applicable.
Item 5. Other Information
During the three months ended December 31, 2023, no director or officer of the Corporation
Item 6. Exhibits
Exhibit No. |
|
Description |
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
|
||
|
|
|
101.INS |
|
Inline XBRL Instance Document – the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document |
101.SCH |
|
Inline XBRL Taxonomy Extension Schema With Embedded Linkbase Documents |
104 |
|
Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
+ Management contract, compensatory plan or arrangement.
* Filed or furnished herewith
38
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
Dated: February 12, 2024
|
ACASTI PHARMA INC. |
||
|
|
|
|
|
By: |
/s/ Prashant Kohli |
|
|
|
Name: Prashant Kohli |
|
|
|
Title: Chief Executive Officer (Principal Executive Officer) |
|
|
|
|
|
|
By: |
/s/ Robert DelAversano |
|
|
|
Name: Robert DelAversano |
|
|
|
Title: Principal Financial Officer (Principal Financial Officer and Principal Accounting Officer) |
|
39